SAPONIFIED CELLULOSE ACETATE
|
Paul Schützenberger discovered that cellulose could be reacted with acetic anhydride to form cellulose acetate. The triacetate is only[citation needed] soluble in chloroform making the method expensive. The discovery that hydrolyzed cellulose acetate is soluble in more polar solvents, like acetone, made production of cellulose acetate fibers cheap and efficient.
|
PRODUCTION
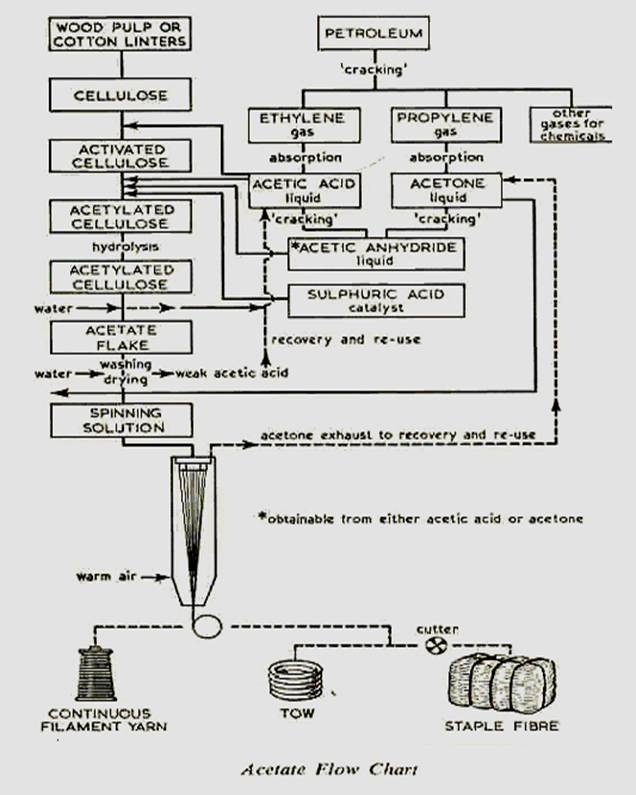
Rayon can be produced from cellulose acetate yarns by saponification. Purified cotton is steeped in glacial acetic acid to make it more reactive. It is then acetylated with excess of glacial acetic acid and acetic anhydride, with sulphuric acid to promote the reaction. The cellulose triacetate formed by acetylation is hydrolysed to convert triacetate to diacetate. The resultant mixture is poured into water which precipitates the cellulose acetate. For spinning it is dissolved in acetone, filtered, deaerated and extruded into hot air which evaporates the solvent. A high degree of orientation can be given to the fiber by drawing because of the fact that cellulose acetate is more plastic in nature. Its fiber cross section is nearly round, but lobed. |