COTTON PREPARATION
|
Contents -
|
Scouring and Bleaching
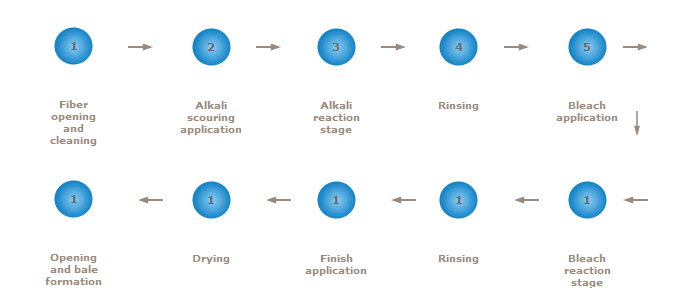
For nonwovens in which absorbency, whiteness and purity are desired, cotton must be scoured and bleached. Two techniques of commercial scouring and bleaching are kier (vat) and continuous (line) processes. Both processes achieve the same results by the same chemical interactions but with different mechanical handling. Figure is a schematic diagram of the sequential operations involved in virgin cotton bleaching for nonwoven applications. 
Scouring is accomplished by saturating the cotton fiber with a caustic soda (sodium hydroxide) solution. This alkali solution is allowed to remain on the fiber at elevated temperatures to speed chemical reactions. During this time, the natural oils and waxes are saponified (converted into soaps), the plant matter is softened, and pectins and other noncellulosic materials are suspended, so that they can be washed away. After a predetermined amount of time to allow for complete scouring, the alkali, saponified waxes, and suspended materials are rinsed away with water.
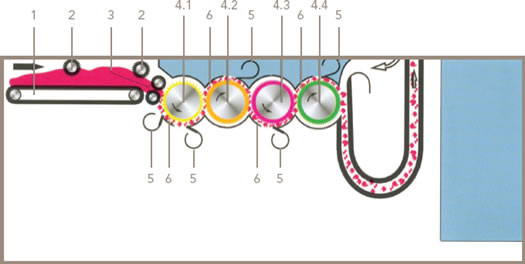
At this point, a bleaching solution is applied to the fiber. A stabilized oxidizing agent, hydrogen peroxide or sodium hypochlorite, is used in the bleaching liquor to whiten the fiber by destroying natural coloring matter. In the United States, hydrogen peroxide is most widely used for bleaching raw cotton in fiber form. The bleaching solution is allowed to remain on the fiber at elevated temperatures for a predetermined amount of time to allow for proper removal of the color bodies, then rinsed away. Cotton bleached with hydrogen peroxide contains no dioxins, because neither lignin nor chlorine is present. After scouring and bleaching, all impurities have been removed, and the cotton fiber is in the form of pure cellulose. If the process is done properly, the fiber will meet the requirements for U.S. Pharmacopoeia purified cotton Opening and Cleaning: Continuous and Kier Bleached Cotton Most of the bleached cotton fiber produced in the United States is processed in large kiers designed to handle several hundred pounds of fiber at once, under high temperature and pressure. Before bleaching, cotton is opened and is generally cleaned to remove non-lint content (trash and dust particles and plant contaminants). The remaining non-lint content should then consist of very small particles that are easily bleached. After bleaching, the wet fiber goes through a moisture extraction operation, and the resulting wet cake is deposited on a spiked apron for opening, followed by drying. The dried fiber then typically passes through some type of opening equipment en route to the bale press. The effectiveness of fiber opening at this stage varies with the following factors:
Figure -- The Cleanomat CXL4, for high-performance cleaning and opening Fiber Finishing and Testing
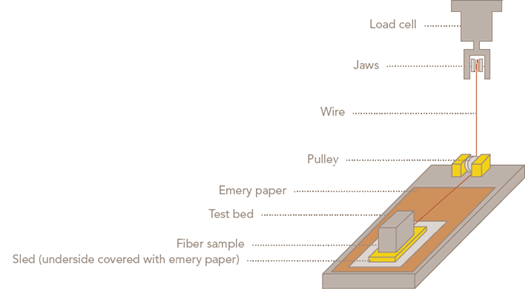
When considering bleached cotton fiber as a stock material for nonwoven webs, it is essential to select the proper fiber finish. Because bleaching removes the natural finish, bleached cotton has high fiber-to-fiber friction. A lubricant and antistatic agent must be added to the fiber to allow it to be processed through the high-production web-forming equipment commonly used for nonwovens. Over the years, such finishes as soap, glycerin and Tween (ethoxylated sorbitan esters) products have been used, with limited success. Cotton Incorporated discovered the value of butoxyethyl stearate (BES), which is now the fiber finish of choice. This chemical finish provides satisfactory lubricity without interfering with absorbency, and it has good acceptability from a toxicity standpoint, which is a more important concern than improved physical properties. A recommended fiber finish system for most bleached cotton includes BES and sodium acetate at add-on levels of 1.0% on weight of fiber (OWF) each. Addition of the salt provides antistatic benefits during mechanical processing, as shown by the trial results. Following is an example of the test procedure:
Figure -- Sled test apparatus for fiber finish evaluation |